Homogeneous catalysis with cAAC-transition metal complexes with low valent, low coordinate metal centers
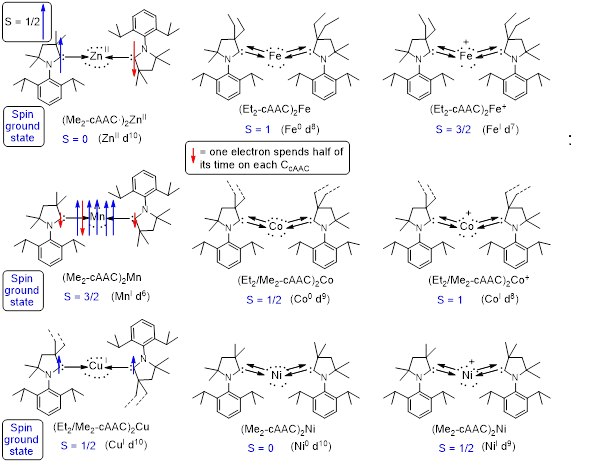
Complexes containing transition metal atoms in different oxidation states are involved in many fundamental processes. They often act as catalysts in various organic transformations. Mechanistic investigations lead to the general conclusion that complexes with higher coordinate metals undergo ligand-dissociation in solution to produce reactive intermediates containing low coordinate metals having available substrate binding sites. Stabilization, characterization, and reactivity studies of such intermediate species can help us to comprehend a more logical and accurate mechanism for the respective reaction. The syntheses of those complexes bearing low valent metal atoms in their zero oxidation states are extremely important and challenging. Complexes containing metal atoms in various oxidation states have been stabilized by different ligands, e.g., carbon monoxide, olefins, phosphines etc. However, it has been observed that with these ligands metal atoms generally adopt higher coordination geometry. Although, N-heterocyclic carbenes (NHCs) are known to stabilize some metal atoms in different oxidation states with low coordination geometry; the successful syntheses of cyclic alkyl amino carbenes (cAACs) opened up a huge possibility to stabilize such species more efficiently.
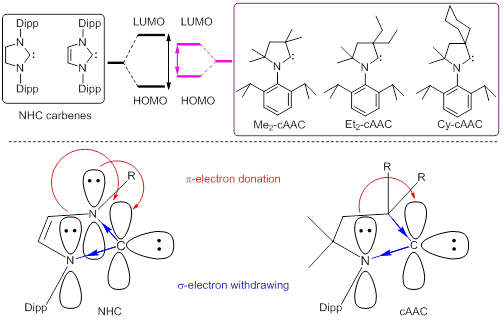
Comparison between NHC (left) and cAAC (right) carbenes.
In cAAC one σ-withdrawing and π-donating nitrogen atom of NHC is replaced by a σ-donating quaternary carbon atom leading to a lower lying LUMO. This makes the acceptance of π-back-donation from the element bound to carbene to the carbon atom of cAAC energetically more advantageous. Experimental results suggested that the carbene carbon of cAAC can utilize the lone pair of electrons on adjacent nitrogen in a more controlled way depending upon the accumulation of electron density on the element bound to it. cAACs form stronger donor-acceptor bonds which provide better stability in the systems. Consequently, cAACs were envisioned to be the superior ligands than NHCs for stabilization of low coordinate metal sites. The cAAC-metal complexes of Zn, Cu, Ni, Co, Fe, Mn [(cAAC)2M-Cl, (cAAC)2M+, (cAAC)2M0] will be employed as efficient catalysts for carrying out unusual organic transformation involving covalent bond activation, oxidative addition and reductive elimination.